
passivated stainless steel
Stainless steel is a versatile material with excellent strength, longevity, and anti-corrosive properties. It has various applications in various industries, from automobile, aerospace, and pharmaceuticals to construction. Generally, stainless steel tends to have natural anti-corrosion properties due to iron molecules preventing rust in specific environments.
However, product designers resort to the passivation of stainless steel to remove iron compounds from the surface of these parts. It reduces the chemical reactivity of stainless steel components by naturally forming an oxide layer on the surface. As a result, it makes them more suitable for different applications.
This article explores in detail the passivation of stainless steel, its benefits, and factors that affect the passivation of stainless steel. Let’s dive in!
What Is Passivation of Stainless Steel?

various passivated stainless steel parts
Passivation of stainless steel is the process of treating stainless steel to make it non-reactive to primary substances such as oxygen and moisture, which can make it rust. Stainless steel passivation occurs by creating a tiny layer of Chromium Oxide (2Cr2O3) on the naked surface of the stainless steel. This process extracts free iron from the metal’s surface with an acid solution leaving behind a surface layer (chromium or nickel) over the underlying steel.
However, the layer left behind helps prevent a reaction between the iron constituent of the stainless steel and the atmospheric compounds. This can cause a deterioration in the material’s property. Mechanical factors, heat, or chemicals can also damage the stainless steel’s corrosion-resistant surface, exposing the item to rusting.
The passivation process serves various purposes in various industries, such as pharmaceuticals, cosmetics, food processing, etc., plagued by safety hazards and massive losses due to equipment corrosion.
Processes in Passivation
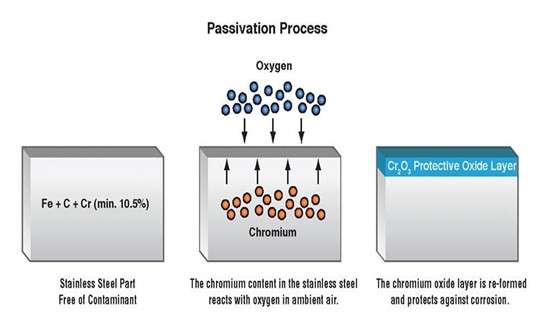
The passivation process
Although passivating stainless steel seems simple, any mishap can cause several complications. Many industries use stainless steel for various sensitive applications, so the slightest processing problem can be costly.
The passivation surface finish involves 3 meticulously taken steps. Here is an overview:
Cleaning Stainless Steel Components
The initial step is ensuring that the stainless steel item is free of contaminants in the passivation process. This presence of organic grease or contaminant on the stainless steel part’s surface will hinder the process’s effectiveness. As a result, you must use an alkaline surface cleanser to extract foreign substances like chemicals, oil particles, and other dirt/contaminants. It is the best cleaning option due to its mildness and effectiveness.
Passivation of Steel Component in Citric Acid Bath or Nitric Acid
At this stage, you must remove the iron compounds from the item’s surface using an acid bath. You must choose between two stainless steel passivation – citric acid and nitric acid.
Traditionally, the citric acid bath is a safe and cost-effective means of executing the passivation process. It is environmentally friendly because it doesn’t create any toxic gas that could harm the environment or the machinist, nor does it require special handling. Although the acid bath passivation had its tendency to grow organic compounds (mold) which hindered its popularity, recent innovations have mitigated these problems.
However, this citric acid immersion/bath process forms a chemical reaction on the stainless component’s surface, irrespective of the procedure. This creates a thin oxide layer with an insignificant amount of iron molecule on the component surface while rest reinstating the metal’s corrosion resistance properties to its natural form.
Nitric acid passivation is a widely used passivator alternative with highly effective molecular reallocation of the molecular structure of the product’s surface and the ability to ensure the longevity of components. Although nitric acid passivation offers these benefits, it requires extended processing time and has several constraints because it is a hazardous material. As a result, it produces toxic gasses which are not eco-friendly; hence, it requires special handling.
In other cases, machinists immerse stainless steel components in nitric acid with sodium dichromate. This solution helps to improve and fasten the passivation process in the case of some particular alloys. However, adding sodium dichromate makes the nitric acid immersion extremely hazardous. As a result, most operators do not use it in passivating stainless steel components.
Testing and Certification
This is the final step in the passivation process, which involves conducting the necessary tests to ensure the pickling and passivation have been done correctly. Although various standards like Mil-Spec, ASTM, and others exist that different industries use worldwide, factors such as target industry, application, and location often determine the appropriate measures.
Try Prolean Now!
What Is the Purpose of Passivating Stainless Steel
Product designers and machinists often passivate for a wide range of reasons. Here are some of them:
- Eradicate contaminants from the product’s surface
- Enhanced surface finish
- Need for reduced system maintenance
- Ensure longer lifespan of products
- Reduce the iron compound in stainless steel to lower its chemical reactivity
Benefits of Passivating Stainless Steel

smoothly passivated tubing
The passivation of stainless steel components comes with a wide range of advantages, including:
Removal of Surface Contaminations
Stainless steel is a material that can be cleaned easily. However, the chemical solution and tool used to extract foreign objects from the surface of the stainless components can cause damage of all kinds to the stainless steel, which can affect its functionality. Passivation helps remove contaminants or debris at a molecular level, leaving the component’s surface undamaged.
Reduced Risk of Contamination in Products
It is critical to keep components used in manufacturing processes with high purity levels, like pharmaceuticals, clean. In addition, iron is unlike Chromium oxide or chrome. It has high chemical reaction levels.
As a result, it can mix with water or other elements to form unwanted compounds resulting in reduced product efficiency or complete damage. More importantly, passivation helps remove compounds other than iron, ensuring that a component is well-cleaned and rid of all contaminants.
Enhanced Resistance to Corrosion
Increased resistance to corrosion is another effect the passivation has on stainless steel components. The passivation process does not only remove contaminants from the surface of the material; it restores and enhances the chromium layer.
As a result, it increases the corrosion resistance level of the material. Passivation treatments can help to remove iron atoms before causing significant damage.
It helps to Save Costs and Avoid Downtime
The passivation process enables you to save yourself the stress of frequent system shutdowns by forming a protective layer on all your stainless steel components. As a result, you can enjoy little-to-no risks of unplanned system shutdowns by preserving the integrity of your stainless steel components. It will also help you save money while significantly increasing your revenue.
Longer System Maintenance Intervals
Putting in place a consistent routine of passivation treatment for reinstating the chemically passive layer of stainless steel components is essential. It will help reduce the need for a total system shutdown due to the need to clean and restore stainless parts in the machines. In addition, it will increase the interval between each scheduled maintenance period.
Furthermore, it is essential to embark on periodic pickling and passivation treatments before using a new vessel. It helps to eliminate any contaminants or foreign objects left over from the manufacturing process.
Passivation Methods for Stainless Steel

passivation of stainless steel part
Chemical Passivation
What is the chemical passivation of stainless steel? Chemical passivation treatment doesn’t change a part’s appearance or surface finish. Instead, it helps to treat stainless steel components by regenerating a thin transparent oxide layer that protects the stainless steel components from corrosion risks by removing the surface contaminants from the base metal.
Electrochemical Passivation
Electrochemical passivation is an electrochemical cleaning done in an electrolytic solution made of chrome to prevent unmanaged oxide growth. This passivation process allows the dissolving or melting of contaminants, usually hard particles found on the stainless steel. It improves the surface finish of stainless steel components, making it suitable for various applications.
Both chemical and electrochemical passivation methods help remove contaminants, increase corrosion resistance, and manage oxide formation on the surface of stainless steel materials. However, the chemical passivation treatment is a non-electrolytic process that forms a very thin transparent oxide coating on the stainless steel protecting it from corrosion.
The electrochemical passivation method erodes hard particle contaminants from the stainless steel surface. It uses electrolytic pickling in passivating the surface and ensuring its corrosion resistance.
Factors Affecting Passivation of Stainless Steel

passivated of large stainless steel part
The passivating of stainless steel parts is similar to controlled corrosion. Here are some factors that may affect the passivation of stainless steel:
Quality of Stainless Steel Material
Although stainless steel has excellent anti-corrosion properties, it can be challenging to handle and attracts lots of dirt and other contaminants that can hinder its passivation. Stainless steel is usually difficult to clean, and spills can stay on the surface longer than expected.
Hence, contaminants in areas that are not cleaned regularly affect the appearance of such surroundings. In addition, machining processes such as welding tend to weaken the stainless steel material due to the high melting point involved in the process.
Cleanliness and Preparation of Surface
The cleanliness and preparation of a stainless steel surface are factors essential to the success of the passivation process. The presence of contaminants, such as cutting oil on a stainless steel part’s surface, can lead to bubbles forming, hindering the process.
As a result, it would be best to use alkaline detergents or degreasers to purify the part’s surface thoroughly. Likewise, you may have to resort to pickling or grinding the surface of a part before passivating it if such parts have thermal oxides due to welding or heat treatment.
Environmental Conditions
Chromium is one of the stainless steel elements that help build a protective layer on its surface, preventing it from rust. However, stainless steel machining processes such as cutting, welding, or grinding can leave behind sulfides, grease, oil, dirt, or other contaminants. These leftover contaminants can cause deterioration in the protective layer or oxides that safeguards the surface from rust.
Conclusion
The passivation process is a necessary fabrication treatment that ensures the longevity of stainless steel products. It strips the stainless steel of the iron compound, which could react with oxygen and cause the surface to rust. Proper passivation of 304 stainless steel products protects such products from rust. More importantly, it is eco-friendly and very safe to practice.
For effective passivation results, it’s important to thoroughly remove all contaminants and grease from the surface of the stainless steel. If you need assistance with your passivation project or require stainless steel machining services, feel free to contact us at ProLean Tech for the best results.
FAQs
What Are the Main Disadvantages of Passivating Stainless Steel?
Stain steel passivation has various disadvantages, as most post-fabrication processes. Passivation doesn’t give the surface of the stainless steel an entirely smooth texture; it only prevents it from corrosion. Likewise, the process requires a pre-cleaning procedure resulting in extended time cycles.
What Are the Common Chemicals Used for Passivating Stainless Steel?
Critic acid, nitric acid, and phosphoric acid are the three significant chemicals commonly used in the passivation process. However, these chemicals have varying strengths, making them suitable for specific applications.



0 Comments