
Bearings with electroplating finish
Surface finishing of CNC machined parts is a vital process in terms of functionality, durability, and appearance. The as-machined finish is not acceptable for most applications and requires post-processing stages.
Numerous surface finishing approaches can be applied for CNC-machined parts based on the material and finishing requirements. The typical finishing process includes sandblasting, Plating, polishing, and painting. And in this article, we will discuss the different plating processes and their importance.
What is a Plating Finish?
The plating process involves covering CNC-machined parts with a thin protective layer of a different metal. The primary purpose of Plating is to alter the properties of the surface, such as roughness, corrosion resistance, wear & tear resistance, and strength. The original properties of the substrate surface (parts or material that need to be plated) might need to be improved for applications, while the Plating can make the parts as desired.
There are four types of the plating process: electroplating, electro-less Plating, mechanical Plating, and immersion plating.
Learn more: electroplating vs electro-less Plating
Electroplating Process

Part with Electroplating finishes
Electroplating use electrolysis to deposit the thin protective layer over the surface of parts. The electrolytic solution is responsible for electro-deposition on the substrate surface. Electroplating involves the technique of depositing a metal onto another through hydrolysis. This method is often utilized to enhance aesthetics or protect the metal from corrosion. Among the various forms are copper, silver, and chromium plating. The image above showcases a component with a stunning electroplating finish, characterized by its dazzlingly smooth surface and commendable anti-corrosion and anti-friction properties.
How does electroplating work?
The working of electroplating is based on the electrolysis principle. It consists of anode, cathode, electrolyte, and DC power sources.
| Cathode | The substrate is connected to the negative terminal of the power source. |
| Anode | The metal desired on the surface is connected to the positive terminal. |
| Electrolytic solution | It contains the ions of metal that need to plate on the substrate surface and also facilitate the oxidation of the anode. |
Let’s understand the working of the electroplating process with an example of copper plating on the zinc surface.
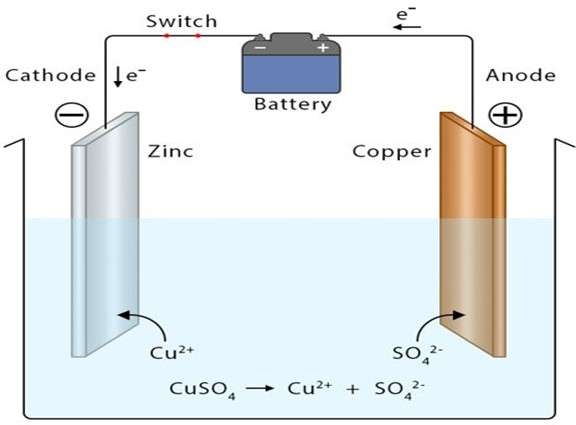
Electroplating of Cu on the Zn surface
Pure copper is connected on the positive terminal (anode), and Zinc on the negative terminal (cathode). Both electrodes are immersed in the electrolytic solution containing copper ions (CuSo4). After the supply of electric current, copper (anode) goes under the oxidation process. The copper ions are dissolved in the electrolytic solution and travel toward the negatively charged zinc substrate. The continuous movement of copper ions (cations) towards Zinc builds a copper layer on the zinc surface.
Thickness, quality, and uniformity of deposition depend on various controlling variables, such as the current magnitude, the distance between two electrodes, and electrolyte temperature.
- Temperature: The right electrolyte temperature facilitates mass transfer and reduces electrolysis resistance.
- Electrodes distance: After the dissolution of the plating metal ion, it travels to the cathode. If the space is too large, it reduces the deposition rate.
- Current magnitude: The magnitude of the current flowing through the circuit decides the oxidation rate of the anode, and it produces the ions for deposition accordingly.
Materials for electroplating
Electroplating can be applied to the CNC-machined parts of any conductive material, including iron, steel, brass, aluminum, Zinc, and silver. However, the latest technology also allows using on the non-metallic parts.
Related: Common Materials for CNC Machining
The following are the most popular materials used to plate various substrates.
- Gold
- Copper
- Silver
- Chromium
- Zinc
- Brass
- Nickel
- Titanium, etc.
The best material for the electroplating depends on the substrate material and properties you want to add. For example, copper is the best option to increase electrical conductivity, while Gold might be the ideal choice for decorating.
Electroless Plating

Parts with electroless-plating finish
As the name suggests, electroless Plating refers to developing a plating layer on the substrate surface without using the electrolysis process. Electroless Plating is also called conversion coating or autocatalytic Plating.
It is the process of creating a protective layer employing the chemical deposition of ions. If you remember, electroplating works well for conductive substrates only. One of the most significant advantages of this approach includes the Plating of non-conductive parts or materials, such as plastics.
How does electroless plating work?
The electroless plating process uses a reducing agent to form the ions of plating metal, which get deposited on the substrate surface through catalyst agents and build a protective layer.
Let’s understand the working of electroless Plating with an example of nickel plating on a steel surface. The nickel sulfate solution (NiSO4 [H2O]6 ) works as the source of nickel ion, and the reducing agent sodium hypophosphite (NaH2PO2 H2O) oxidize the nickel sulfate to create the ions of nickel, which get deposited on the steel surface.
Other solutions like tri-ammonium citrate [(NH4)3C6H5O7)] are also used in the process as the A buffer or mild complex agent (Brand, 2014)
Materials for electroless Plating
Copper, nickel, silver, titanium, and other metals can plate many substrates, including metallic and non-metallic parts. The appropriate reducing agent, complex agent, and buffer solution are crucial to obtain the quality result.
Immersion Plating
Immersion plating refers to coating a substrate with a thin protective layer of another metal by dipping it into the solution of that metal. The primary requirement for immersion plating is that the substrate metal needs to be less noble than the plating metal.
It creates a thin protective layer as compared to electroless and electroplating. This plating approach is beneficial to make parts corrosion resistive, increase hardness, and alter the electrical conductivity.
How does immersion plating work?
The immersion plating process uses a solution that contains the coating metal. The coating metal solution displaces the substrate metal ions from the surface. It is also known as displacement plating since one metal displaces to form a coating layer.
The phenomenon happens because of the higher oxidation potential of displaced metal. Let’s understand an example of copper plating on a steel surface. When the steel is immersed in the solution containing copper ions, the iron displaces the copper from the solution since the copper has more oxidation potential than the iron.
Fe + Cu2+ = Cu + Fe2+ 【by Stanley Hirsch, Charles Rosenstein]
The deposition of coating metal stops when the surface of the base metal is completely covered. Plating thickness gets influenced by different variables, such as temperature, reaction rate, and surface quality.
Materials for immersion plating
Many metals and alloys, including iron, steel, copper, Zinc, and aluminum, are suitable for immersion plating. In industrial applications, Gold, Silver, Copper, Tin, Zinc, Platinum, Rhodium, and Ruthenium are the most used metals to create a protective coating.
Mechanical Plating

Mechanical-plated parts
Mechanical Plating is done by dipping the base material into the plating medium. It is a cost-effective approach for coating many parts quickly, with more advantages than galvanization. This process requires clean, contamination-free metal parts and can be operated at room temperature. However, a slightly high temperature can speed up the process.
How does mechanical plating work?
The base metal is dipped into the plating drum containing the powder/solution of the coating metal. Other materials are also in the plating drum, such as glass balls and silicon particles, which hammer the coating powder into the substrate surface once the drum starts to rotate.
Materials for mechanical Plating
Zinc, cadmium, tin, copper, and aluminum are the common material used to coat the substrate surface with a mechanical plating approach. However, Zinc is a widely used metal among these.
Differences Between Coating And Plating
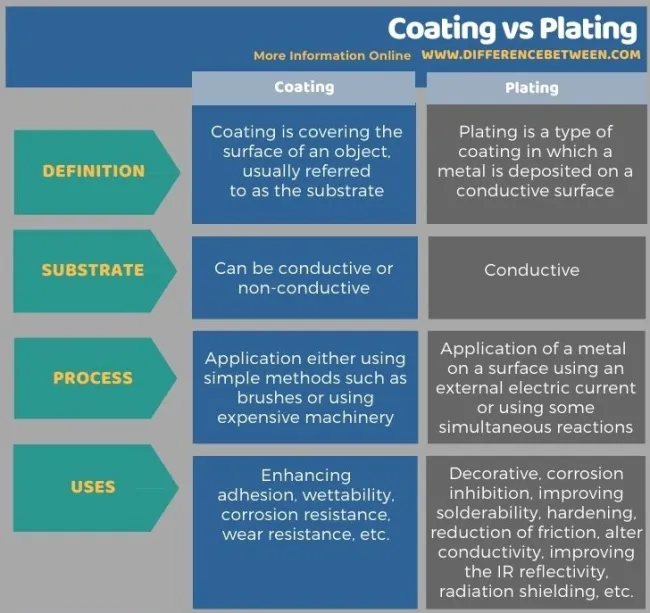
Differences Between Coating And Plating
Coating and plating are two different surface finishing techniques used in various industries, including metalworking and electronics.
Coating refers to the process of applying a thin layer of material onto a substrate to provide a desired finish, protection, or performance enhancement. Coatings can be applied using various methods, including brushing, spraying, dipping, or electrostatic application. Examples of coatings include paint, powder coating, and anodizing.
Plating involves the deposition of a thin layer of metal onto a substrate. Plating is typically performed through electroplating, where a metal object is submerged in a solution containing ions of the metal to be deposited. An electric current is then passed through the solution, causing the metal ions to bond to the surface of the object. Plating is used to enhance the appearance, corrosion resistance, and conductivity of the substrate.
Importance of Metal Plating
We discussed four metal plating approaches for surface finishing. The primary benefits of Plating include corrosion resistance, conductivity improvement, and aesthetic use. However, each plating process has unique advantages in the surface finishing of CNC-machined parts. Let’s discuss the key importance of plating finish.
- Protection from Corrosion
It is the sole purpose of any plating finish on the CNC machined parts. Developing a protective layer of metal with superior corrosion resistance makes parts resistant to corrosion, even in a moist environment.
- Aesthetic appeal
Aesthetic appeal is essential in the manufacturing industry, from simple mechanical tools to well-designed jewelry that needs an appealing appearance. A plating finish can be applied on metallic and non-metallic parts to improve their appearance. For example, zinc plating on steel provides a shiny silver surface.
- Wear resistance
Coating a protective layer of hard metal on the surface of base materials improves the hardness and makes it resistant to wear & tear. For example, aluminum becomes more resistant to starches and wear after plating Zinc on the surface.
- Improvement of Electrical & Thermal conductivity
Plating is the most efficient and economical method for improving various parts’ thermal and electrical conductivity. For example, copper plating is used in electronic parts to make them highly electrically conductive.
- Uniform surface finish
Plating offers a uniform surface finish, and the thickness of the plating layer developed consistently across the substrate surface. Electro-plating allows the creation of the plating material layer with the desired consistency all over the surface.
Application of Plating
| Decorative plating | To add an aesthetically pleasing layer to jewelry, cutlery, and other decorative items. |
| Protective plating | To protect against corrosion, wear, and tear, and to improve the overall durability of metal parts used in various industries such as automotive, aerospace, and electronics. |
| Electroplating | To improve the electrical conductivity of metal parts used in electronics and electrical equipment. |
| Biomedical plating | To repair or replace damaged bones or tissues in medical procedures such as orthopedic surgery. |
| Hard plating | To increase the hardness and wear resistance of metal parts used in various industrial applications. |
| Plating on plastics |
To enhance the physical and/or electrical properties of plastic parts used in various industries such as automotive, electronics, and consumer goods. |
Conclusion
Plating is undoubtedly one of the most effective and economical surface finishing approaches for a wide range of materials. Based on the specification of the required finish, various metals can be plated into parts created from CNC machining or other manufacturing methods.
Four distinct plating methods can be applied to parts or materials to modify their surface properties. The right method, equipment, and expertise are paramount to achieve optimal plating results. Prolean stands out as your go-to surface plating experts, offering various surface finishing options, including the coveted plating finish. If you’re in search of on-demand manufacturing services to bring your design to life, trust Prolean, your surface plating experts, to be your most valuable collaborative partner.
FAQ’s
What are the advantages of plating surface finish?
Plating is the cost-effective approach to customize the surface properties of material or parts. It offers excellent corrosion resistance, wear & tear resistance, durability, and a glossy appearance.
What is the difference between electroless and immersion Plating?
The fundamental difference between immersion and electroless Plating is that it does not utilize any reducing agent or catalytic solution for deposition.
Which plating approach is best for my CNC -machining project?
It depends on various factors, such as material type, required specifications, cost, end-applications of parts, and many more.
Bibliography
- Brand, A. (2014). Plated Nickel-CoppPlated Nickel-Copper Contacts on c-Si: from Microelectronic Processing to Cost Effective Silicon Solar Cell Production. ResearchGate.
- by Stanley Hirsch and Charles Rosenstein. (n.d.). IMMERSION PLATING. NY: Leeam Consultants Ltd., New Rochelle.



0 Comments